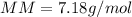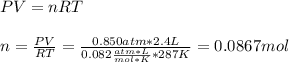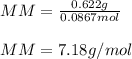Chemistry, 04.08.2020 23:01 jamarengle2
Part 1. Determine the molar mass of a 0.622-gram sample of gas having a volume of 2.4 L at 287 K and 0.850 atm. Show your work. Part 2. If this sample was placed under extremely low temperature, describe how the actual volume would compare to the predicted volume. Explain your answer.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1

Chemistry, 22.06.2019 19:30
Awoman's basketball has a circumference between 28.5 and 29.0 inches and a maximum weight of 20 ounces (two significant figures). what are these specifications in units of centimeters and grams?
Answers: 2

Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3

Chemistry, 23.06.2019 02:00
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2
You know the right answer?
Part 1. Determine the molar mass of a 0.622-gram sample of gas having a volume of 2.4 L at 287 K and...
Questions




Computers and Technology, 16.07.2019 06:30


Mathematics, 16.07.2019 06:30



Chemistry, 16.07.2019 06:30

English, 16.07.2019 06:30

History, 16.07.2019 06:30


History, 16.07.2019 06:30


History, 16.07.2019 06:30

Business, 16.07.2019 06:30


Social Studies, 16.07.2019 06:30


English, 16.07.2019 06:30