
Chemistry, 12.08.2020 06:01 munziruddin204
Consider the following: Li(s) + ½ I₂(g) --> LiI(s) ΔH = –292 kJ. LiI(s) has a lattice energy of –753 kJ/mol. The ionization energy of Li(g) is 520 kJ/mol, the bond energy of I₂(g) is 151 kJ/mol, and the electron affinity of I(g) is –295 kJ/mol. Use these data to determine the heat of sublimation of Li(s).

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
9. write the chemical equation for the following word equations. include symbols for physical states in the equation. a. solid zinc sulfide + oxygen gas -> solid zinc oxide + sulfur dioxide gas b. aqueous hydrochloric acid + aqueous barium hydroxide -> aqueous barium chloride + water
Answers: 1

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 13:00
Imagine that you push on a large rock. at what point does your effort change the rock’s mechanical energy?
Answers: 1

Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1
You know the right answer?
Consider the following: Li(s) + ½ I₂(g) --> LiI(s) ΔH = –292 kJ. LiI(s) has a lattice energy of –...
Questions

English, 05.01.2021 22:20

Social Studies, 05.01.2021 22:20

Geography, 05.01.2021 22:20

History, 05.01.2021 22:20

Mathematics, 05.01.2021 22:20

Mathematics, 05.01.2021 22:20


Mathematics, 05.01.2021 22:20

English, 05.01.2021 22:20

English, 05.01.2021 22:20

Arts, 05.01.2021 22:20


Spanish, 05.01.2021 22:20

Mathematics, 05.01.2021 22:20

History, 05.01.2021 22:20



Physics, 05.01.2021 22:20

Mathematics, 05.01.2021 22:20

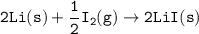
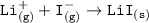
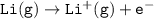
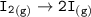
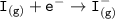
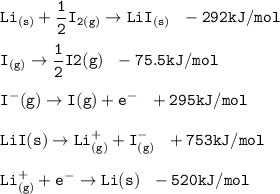
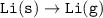 = (-292 +(-75.5)+295+753+(-520)) kJ/mol
= (-292 +(-75.5)+295+753+(-520)) kJ/mol

