
Chemistry, 12.08.2020 06:01 valenzueladomipay09u
Calculate the amount of heat that must be absorbed by 10.0 g of ice at –20°C to convert it to liquid water at 60.0°C. Given: specific heat (ice) = 2.1 J/g·°C; specific heat (water) = 4.18 J/g·°C; ΔH fus = 6.0 kJ/mol.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3

Chemistry, 22.06.2019 22:30
The diagram shows the relationship between scientific disciplines.the names of some scientific disciplines have been removed from the boxes. which scientific discipline belongs in the blue box? a.physics b.biology c.chemistry d.metallurgy
Answers: 2

You know the right answer?
Calculate the amount of heat that must be absorbed by 10.0 g of ice at –20°C to convert it to liquid...
Questions



Mathematics, 26.06.2019 14:30

Chemistry, 26.06.2019 14:30

Mathematics, 26.06.2019 14:30

Mathematics, 26.06.2019 14:30

Biology, 26.06.2019 14:30







Chemistry, 26.06.2019 14:30

Mathematics, 26.06.2019 14:30

Mathematics, 26.06.2019 14:30

Mathematics, 26.06.2019 14:30

Mathematics, 26.06.2019 14:30


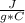 and ΔT=0 C - (-20 C)= 20 C
and ΔT=0 C - (-20 C)= 20 C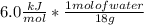 = 3.333 kJ= 3,333 J (being kJ=1,000 J)
= 3.333 kJ= 3,333 J (being kJ=1,000 J)

