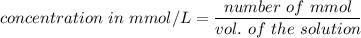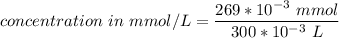Chemistry, 05.08.2020 17:01 NathanChavez4313
A chemist prepares a solution of silver(I) nitrate(AgNO3) by measuring out 269. mu mol of silver(I) nitrate into a 300. mL volumetric flask and filling the flask to the mark with water. Calculate the concentration in mmol/L of the chemist's silver(I) nitrate solution.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1

Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3
You know the right answer?
A chemist prepares a solution of silver(I) nitrate(AgNO3) by measuring out 269. mu mol of silver(I)...
Questions


Biology, 19.10.2021 14:00

Business, 19.10.2021 14:00


Physics, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00


Social Studies, 19.10.2021 14:00

English, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00

Health, 19.10.2021 14:00


Computers and Technology, 19.10.2021 14:00

Mathematics, 19.10.2021 14:00

Physics, 19.10.2021 14:00

Health, 19.10.2021 14:00




Spanish, 19.10.2021 14:00