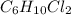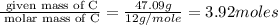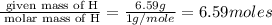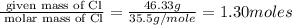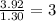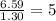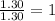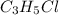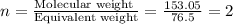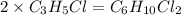Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

You know the right answer?
Compound has a molar mass of and the following composition: elementmass % carbon47.09% hydrogen6.59%...
Questions

Mathematics, 03.08.2020 14:01


Spanish, 03.08.2020 14:01


English, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01

English, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01


Biology, 03.08.2020 14:01



Mathematics, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01

History, 03.08.2020 14:01




Mathematics, 03.08.2020 14:01

Mathematics, 03.08.2020 14:01