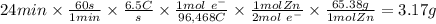Chemistry, 12.08.2020 06:01 sparkybig12
Zinc is used as a coating for steel to protect the steel from environmental corrosion. If a piece of steel is submerged in an electrolysis bath for 24 minutes with a current of 6.5 Amps, how many grams of zinc will be plated out? The molecular weight of Zn is 65.38, and Zn+2 + 2e– → Zn. Question 7 options: A) 3.17 g of Zn B) 1.09 g of Zn C) 6.34 g of Zn D) 12.68 g of Zn

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 03:30
Each pair of clay balls represents to planetesimals if each plane test molluscum pound of the same material and is separated by the same distance which pair experiences the greatest gravitational attraction
Answers: 2

Chemistry, 22.06.2019 20:00
How are the terms group and period used on the periodic table
Answers: 1

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3
You know the right answer?
Zinc is used as a coating for steel to protect the steel from environmental corrosion. If a piece of...
Questions

Business, 08.03.2021 20:30

Biology, 08.03.2021 20:30



Physics, 08.03.2021 20:30

Arts, 08.03.2021 20:30

Chemistry, 08.03.2021 20:30


Mathematics, 08.03.2021 20:30

Health, 08.03.2021 20:30


English, 08.03.2021 20:30


Social Studies, 08.03.2021 20:30

History, 08.03.2021 20:30

History, 08.03.2021 20:30

Mathematics, 08.03.2021 20:30



Mathematics, 08.03.2021 20:30