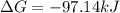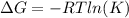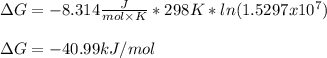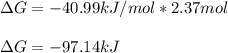A student determines the value of the equilibrium constant to be 1.5297 x 107 for the following reaction: HBr(g) + 1/2 Cl2(g) --> HCl(g) +1/2 Br2(g) Based on this value of Keq, calculate the Gibbs free energy change for the reaction of 2.37 moles of HBr(g) at standard conditions at 298 K.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
The wave shown on the electromagnetic spectrum disturb the medium it passes through a)different frequency. b)the same frequency .
Answers: 2

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2
You know the right answer?
A student determines the value of the equilibrium constant to be 1.5297 x 107 for the following reac...
Questions

Mathematics, 18.10.2021 07:50


Advanced Placement (AP), 18.10.2021 07:50

Mathematics, 18.10.2021 07:50




Mathematics, 18.10.2021 07:50

Mathematics, 18.10.2021 07:50

Engineering, 18.10.2021 07:50



Computers and Technology, 18.10.2021 07:50

Mathematics, 18.10.2021 07:50


Biology, 18.10.2021 07:50



Mathematics, 18.10.2021 08:00

Spanish, 18.10.2021 08:00