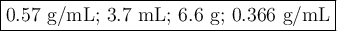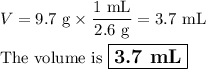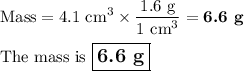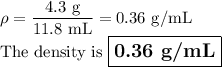Chemistry, 12.08.2020 06:01 naomicervero
Find the density if the volume is 15 mL and the mass is 8.6 g. (5 pts)
Find the volume if the density is 2.6 g/mL and the mass is 9.7 g.(5 pts)
Find the mass if the density is 1.6 g/cm3 and the volume is 4.1 cm3 (5 pts)
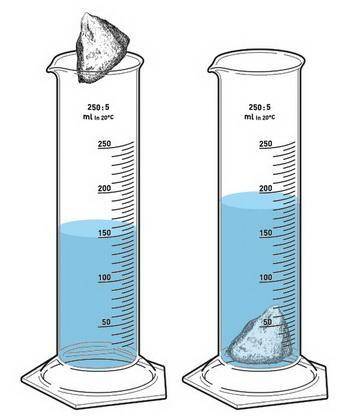
Find the density if the initial volume of water is 12.8 mL, the final volume is 24.6 mL and the mass of the object is 4.3 g. Make a drawing to show the water displacement using a graduated cylinder. (gdoc, gdraw)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Infants born with severe respiratory problems are sometimes given liquid ventilation: they breathe a liquid that can dissolve more oxygen than air can hold. one of these liquids is a fluorinated compound, cf3(cf2)7br. the solubility of oxygen in this liquid is 66 mlo2 per 100 ml liquid. in contrast, air is 21 % oxygen by volume. calculate the moles of o2 present in an infant's lungs (volume: 12 ml ) if the infant takes a full breath of air. assume a pressure of 1 atm in the lungs.
Answers: 1

Chemistry, 21.06.2019 16:30
How does decreasing the gas volume affect the pressure of a gas?
Answers: 1

Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1

Chemistry, 23.06.2019 01:00
The primary products of complete combustion of fossil fuels are a. carbon dioxide and water b. methane and water c. carbon monoxide and water d. carbon dioxide and carbon monoxide
Answers: 1
You know the right answer?
Find the density if the volume is 15 mL and the mass is 8.6 g. (5 pts)
Find the volume if the densi...
Questions


Social Studies, 25.02.2021 19:30



Mathematics, 25.02.2021 19:30



Mathematics, 25.02.2021 19:30


Mathematics, 25.02.2021 19:30


Mathematics, 25.02.2021 19:30

Biology, 25.02.2021 19:30

Computers and Technology, 25.02.2021 19:30


History, 25.02.2021 19:30

Mathematics, 25.02.2021 19:30