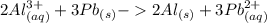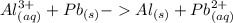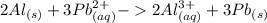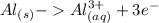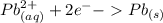Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 09:00
Astudent is asked to identify and element that is pale yellow brittle solid and does not conduct electricity. at which location in this periodic table would the element most likely be found?
Answers: 2

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

Chemistry, 23.06.2019 13:50
Use the periodic table and your knowledge of isotopes to complete these statements. when polonium-210 emits an alpha particle, the child isotope has an atomic mass of 1-131 undergoes beta-minus decay. the chemical symbol for the new element is fluorine-18 undergoes beta-plus decay. the child isotope has an atomic mass of done intro donne
Answers: 1
You know the right answer?
Which balanced redox reaction is occurring in the voltaic cell represented by the notation of A l (...
Questions

Geography, 10.10.2019 14:50


History, 10.10.2019 14:50

Health, 10.10.2019 14:50

History, 10.10.2019 14:50


Mathematics, 10.10.2019 14:50

Social Studies, 10.10.2019 14:50





Biology, 10.10.2019 14:50

Mathematics, 10.10.2019 14:50

Mathematics, 10.10.2019 14:50

Biology, 10.10.2019 14:50



Computers and Technology, 10.10.2019 14:50

Spanish, 10.10.2019 14:50

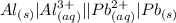 ?
?