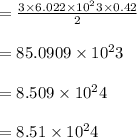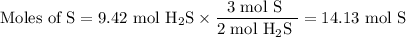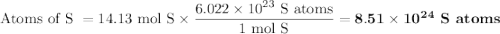Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Of the groups of elements below, which are most likely to gain electrons to become anions? a. alkali metal b. boron group c. halogen d. transition metal
Answers: 2

Chemistry, 22.06.2019 02:00
Which of the following happens during cell division? (a) energy is created (b) waste is eliminated (c) carbon dioxide is released (d) damaged cells are replaced
Answers: 1

Chemistry, 22.06.2019 04:00
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2
You know the right answer?
How many sulfur atoms are generated when 9.42 moles of H2S react according to the following equation...
Questions





Mathematics, 24.01.2021 21:10

Social Studies, 24.01.2021 21:10


Biology, 24.01.2021 21:10




English, 24.01.2021 21:10








 .
.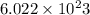 no of atoms & molecules. The calculation is as follows:
no of atoms & molecules. The calculation is as follows: