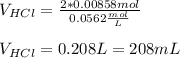Chemistry, 14.08.2020 01:01 Lorettareneemills
People take antacids, such as milk of magnesia, to reduce the discomfort of acid stomach or heartburn. The recommended dose of milk of magnesia is 1 teaspoon, which contains 500 mg of Mg(OH)2. What volume of HCl solution with a pH of 1.25 can be neutralized by 1 dose of milk of magnesia

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 01:30
Sulfuric acid (a component of acid rain) reacts with limestone (calcium carbonate) to produce calcium sulfate and carbon dioxide. this damages buildings and statues made of limestone. which solution of sulfuric acid will damage these structures more quickly? a. 0.001% b. 0.005% c. 0.010% d. 0.015%
Answers: 3

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
People take antacids, such as milk of magnesia, to reduce the discomfort of acid stomach or heartbur...
Questions


Mathematics, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

Social Studies, 25.01.2021 21:20

Medicine, 25.01.2021 21:20

Chemistry, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

Chemistry, 25.01.2021 21:20

SAT, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20




History, 25.01.2021 21:20

Mathematics, 25.01.2021 21:20

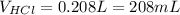
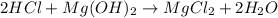

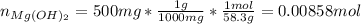
![[H^+]=[HCl]=10^{-pH}=10^{-1.25}=0.0562M](/tpl/images/0721/9030/16c77.png)
![[HCl]*V_{HCl}=2*n_{Mg(OH)_2}](/tpl/images/0721/9030/0b27d.png)