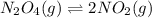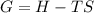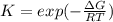Chemistry, 14.08.2020 04:01 sillslola816oxb5h7
If we represent the equilibrium as:...N2O4(g) 2 NO2(g) We can conclude that: 1. This reaction is: A. Exothermic B. Endothermic C. Neutral D. More information is needed to answer this question. 2. When the temperature is increased the equilibrium constant, K: A. Increases B. Decreases C. Remains the same D. More information is needed to answer this question. 3. When the temperature is increased the equilibrium concentration of NO2: A. Increases B. Decreases C. Remains the same D. More information is needed to answer this question.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Which of the following best explains why the end of a spoon sticking out of a cup of hot water also gets hot? question 7 options: the heat from the hot water is conducted through the spoon handle the hot water heats the air surrounding the upper part of the spoon. the hot water causes a physical change in the spoon handle. the hot water causes a chemical reaction to take place in the spoon.
Answers: 2



Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1
You know the right answer?
If we represent the equilibrium as:...N2O4(g) 2 NO2(g) We can conclude that: 1. This reaction is: A....
Questions

English, 16.08.2021 15:00

Biology, 16.08.2021 15:00

English, 16.08.2021 15:00



Physics, 16.08.2021 15:00

Mathematics, 16.08.2021 15:00

Social Studies, 16.08.2021 15:00


Biology, 16.08.2021 15:00

Mathematics, 16.08.2021 15:00



Mathematics, 16.08.2021 15:00

Social Studies, 16.08.2021 15:00

English, 16.08.2021 15:00


Mathematics, 16.08.2021 15:00

Biology, 16.08.2021 15:00