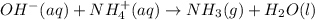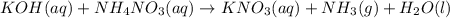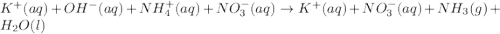Chemistry, 13.08.2020 18:01 SMURFETTE86
Consider the reaction when aqueous solutions of potassium hydroxide and ammonium nitrate are combined. The net ionic equation for this reaction is:

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 12:00
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom
Answers: 1
You know the right answer?
Consider the reaction when aqueous solutions of potassium hydroxide and ammonium nitrate are combine...
Questions

Mathematics, 08.03.2021 18:00





Chemistry, 08.03.2021 18:00

Social Studies, 08.03.2021 18:00




Biology, 08.03.2021 18:00




English, 08.03.2021 18:00

Arts, 08.03.2021 18:00



History, 08.03.2021 18:00

History, 08.03.2021 18:00