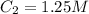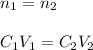Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:00
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3


You know the right answer?
2. You deposit the 500 ul from #1 into a solution with a final volume of 1200 uL. What is the final...
Questions

Mathematics, 28.01.2020 11:31


History, 28.01.2020 11:31

Social Studies, 28.01.2020 11:31


Biology, 28.01.2020 11:31


Mathematics, 28.01.2020 11:31


Mathematics, 28.01.2020 11:31

English, 28.01.2020 11:31




Mathematics, 28.01.2020 11:31


Mathematics, 28.01.2020 11:31

Mathematics, 28.01.2020 11:31