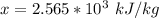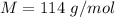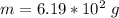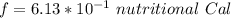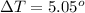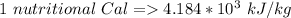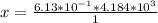A 0.0447−mol sample of a nutrient substance, with a formula weight of 114 g/mol, is burned in a bomb calorimeter containing 6.19 × 102 g H2O. Given that the fuel value is 6.13 × 10−1 in nutritional Cal when the temperature of the water is increased by 5.05°C, what is the fuel value in kJ in scientific notation?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

You know the right answer?
A 0.0447−mol sample of a nutrient substance, with a formula weight of 114 g/mol, is burned in a bomb...
Questions






English, 24.09.2019 21:00

Social Studies, 24.09.2019 21:00


Mathematics, 24.09.2019 21:00

Mathematics, 24.09.2019 21:00


Computers and Technology, 24.09.2019 21:00

Chemistry, 24.09.2019 21:00

Biology, 24.09.2019 21:00


Business, 24.09.2019 21:00


English, 24.09.2019 21:00

Physics, 24.09.2019 21:00