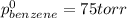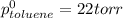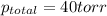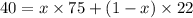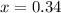Chemistry, 18.08.2020 21:01 zitterkoph
At 20°C the vapor pressure of benzene (C6H6) is 75 torr, and that of toluene (C7H8) is 22 torr. Assume that benzene and toluene form an ideal solution. What is the composition in mole fractions of a solution that has a vapor pressure of 40. torr at 20°C?

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2

Chemistry, 23.06.2019 02:30
What type of energy conversion occurs when you place your feet near the fire place and they become warm
Answers: 1

Chemistry, 23.06.2019 02:40
How can a mixture of salt water be separated into salt and water
Answers: 1
You know the right answer?
At 20°C the vapor pressure of benzene (C6H6) is 75 torr, and that of toluene (C7H8) is 22 torr. Assu...
Questions


Mathematics, 25.03.2022 19:50




Chemistry, 25.03.2022 19:50

Biology, 25.03.2022 19:50


SAT, 25.03.2022 20:10


Mathematics, 25.03.2022 20:20

Mathematics, 25.03.2022 20:20


Computers and Technology, 25.03.2022 20:20

English, 25.03.2022 20:30

Chemistry, 25.03.2022 20:30




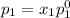 and
and 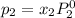
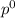 = pressure in the pure state
= pressure in the pure state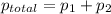
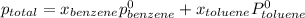
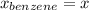 ,
,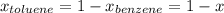 ,
,