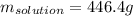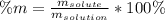How many grams of a 14.0% (w/w) sugar solution contain 62.5
of sugar?...

Chemistry, 24.08.2020 23:01 yesman1288
How many grams of a 14.0% (w/w) sugar solution contain 62.5
of sugar?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3
You know the right answer?
Questions

History, 23.09.2019 12:30



Mathematics, 23.09.2019 12:50

Biology, 23.09.2019 12:50



History, 23.09.2019 12:50


Social Studies, 23.09.2019 12:50

Biology, 23.09.2019 12:50


Biology, 23.09.2019 12:50




Mathematics, 23.09.2019 12:50

Mathematics, 23.09.2019 12:50