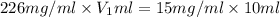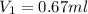Chemistry, 27.08.2020 23:01 joelpimentel
You have a stock solution that is 226mg/mL and you need 10mL of a working solution that is 15mg/mL. What is the volume of stock solution will you need to dilute to have 10mL of working solution at the above concentration. State your answer to two decimals.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Smog is the term used to describe the combination of fog and smoke
Answers: 1

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
You know the right answer?
You have a stock solution that is 226mg/mL and you need 10mL of a working solution that is 15mg/mL....
Questions



Social Studies, 10.10.2019 20:10


Mathematics, 10.10.2019 20:10



History, 10.10.2019 20:10

Biology, 10.10.2019 20:10


Mathematics, 10.10.2019 20:10


Chemistry, 10.10.2019 20:10



English, 10.10.2019 20:10




Mathematics, 10.10.2019 20:10

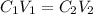
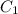 = concentration of stock solution = 226 mg/ml
= concentration of stock solution = 226 mg/ml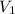 = volume of stock solution = ?
= volume of stock solution = ?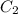 = concentration of working solution= 15 mg/ml
= concentration of working solution= 15 mg/ml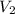 = volume of working solution= 10 ml
= volume of working solution= 10 ml