
Chemistry, 31.08.2020 02:01 dylancasebere
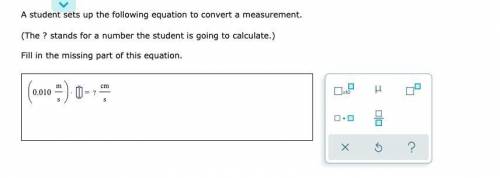
A student sets up the following equation to convert a measurement. (The ? stands for a number the student is going to calculate.) Fill in the missing part of this equation.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2
You know the right answer?
A student sets up the following equation to convert a measurement. (The ? stands for a number the st...
Questions

Physics, 06.11.2020 18:00




Mathematics, 06.11.2020 18:00


Mathematics, 06.11.2020 18:00



Health, 06.11.2020 18:00


Chemistry, 06.11.2020 18:00

Mathematics, 06.11.2020 18:00


Mathematics, 06.11.2020 18:00


Social Studies, 06.11.2020 18:00

Mathematics, 06.11.2020 18:00




