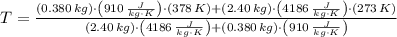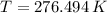Chemistry, 01.09.2020 02:01 davidcortez27
A 0.380 kg sample of aluminum (with a specific heat of 910.0 J/(kg x K)) is heated to 378 K and then placed in 2.40 kg of water that is at 293 K. If the system is left to reach its equilibrium state, what will the equilibrium temperature be? Assume that no thermal energy is lost to the external environment. Use 4.186 J/(g x K) as the specific heat of water.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1

Chemistry, 23.06.2019 02:30
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1
You know the right answer?
A 0.380 kg sample of aluminum (with a specific heat of 910.0 J/(kg x K)) is heated to 378 K and then...
Questions



Mathematics, 14.12.2021 03:40





Mathematics, 14.12.2021 03:40

Mathematics, 14.12.2021 03:40

Chemistry, 14.12.2021 03:40


Mathematics, 14.12.2021 03:40



Physics, 14.12.2021 03:40

Computers and Technology, 14.12.2021 03:40

Social Studies, 14.12.2021 03:40



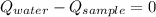
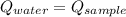
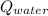 - Heat received by water, measured in joules.
- Heat received by water, measured in joules.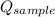 - Heat released by the sample of aluminium, measured in joules.
- Heat released by the sample of aluminium, measured in joules.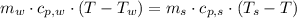
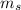 ,
, 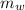 - Mass of water and the sample of aluminium, measured in kilograms.
- Mass of water and the sample of aluminium, measured in kilograms.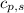 ,
, 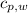 - Specific heats of the sample of aluminium and water, measured in joules per kilogram-Kelvin.
- Specific heats of the sample of aluminium and water, measured in joules per kilogram-Kelvin.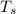 ,
, 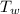 - Initial temperatures of the sample of aluminium and water, measured in Kelvin.
- Initial temperatures of the sample of aluminium and water, measured in Kelvin.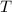 - Temperature which system reaches thermal equilibrium, measured in Kelvin.
- Temperature which system reaches thermal equilibrium, measured in Kelvin.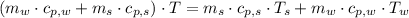
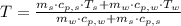
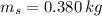 ,
, 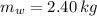 ,
, 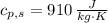 ,
, 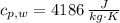 ,
, 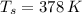 and
and 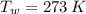 , the final temperature of the system is:
, the final temperature of the system is: