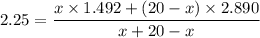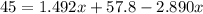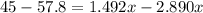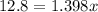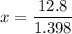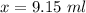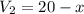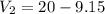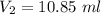Suppose now that you wanted to determine the density of a small crystal to confirm that it is graphite. From the literature, you know that graphite has a density of 2.25 g/cm^3. How would you prepare 20.0 mL of the liquid mixture having that density from pure samples of CHCl3 (d = 1.492 g/mL) and CHBr3 (d = 2.890 g/mL)? (Note: 1 mL = 1 cm^3.)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 17:10
For which one of the following reactions is the value of δh° rxn equal to δh° f for the product? a. 2 h2 (g) + o2 (g) → 2 h2o (l) b. n2 (g) + o2 (g) → 2 no (g) c. 2 h2 (g) + o2 (g) → 2 h2o (g) d. h2o (l) + 1/2 o2 (g) → h2o2 (l) e. none of the above
Answers: 1

Chemistry, 23.06.2019 00:30
Which of the following best describes technology a. something created for only scientists to use b.the method of thinking that scientists use. c.the application of engineering to create useful products. c. a scientific idea
Answers: 1

Chemistry, 23.06.2019 01:00
What is the chemical name of the compound ti2o3? use the list of polyatomic ions and the periodic table to you answer.
Answers: 1

Chemistry, 23.06.2019 04:40
[01.07]what is the answer to the problem: 101 g + 25.01 g + 5.05 g? 131.06 g 131.1 g 131 g 130 g
Answers: 1
You know the right answer?
Suppose now that you wanted to determine the density of a small crystal to confirm that it is graphi...
Questions

Chemistry, 20.10.2020 09:01


Health, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01



Chemistry, 20.10.2020 09:01


English, 20.10.2020 09:01



Mathematics, 20.10.2020 09:01

Mathematics, 20.10.2020 09:01

Advanced Placement (AP), 20.10.2020 09:01

Mathematics, 20.10.2020 09:01

World Languages, 20.10.2020 09:01

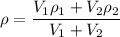
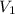 = volume of first compound
= volume of first compound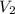 = volume of second compound
= volume of second compound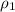 =density of first compound
=density of first compound