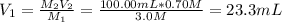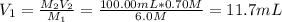Chemistry, 01.09.2020 23:01 haleyblu6351
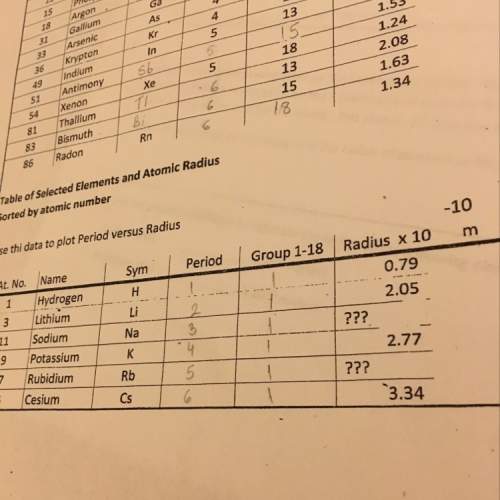
Use a volumetric flask to prepare 100.00 mL of 0.70M HCl solution. Do this by diluting either one of the stock hydrochloric acid solutions that have been set out for you to use (the stock solutions are 3.0M and 6.0M HCl). (WARNING! Concentrated HCl is caustic and will cause burns. Handle carefully.) Dispose of any excess HCl in the waste container.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:10
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1

Chemistry, 22.06.2019 00:30
This element exists in adundance in the sun.explain how you would go about capturing sunlight.would this captured sunlight contain any of the element?
Answers: 1

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1
You know the right answer?
Use a volumetric flask to prepare 100.00 mL of 0.70M HCl solution. Do this by diluting either one of...
Questions

Computers and Technology, 08.12.2019 13:31

History, 08.12.2019 13:31

Chemistry, 08.12.2019 13:31







Social Studies, 08.12.2019 13:31

Mathematics, 08.12.2019 13:31

Social Studies, 08.12.2019 13:31

Biology, 08.12.2019 13:31


Physics, 08.12.2019 13:31

Advanced Placement (AP), 08.12.2019 13:31

Mathematics, 08.12.2019 13:31

Mathematics, 08.12.2019 13:31

Mathematics, 08.12.2019 13:31

History, 08.12.2019 13:31