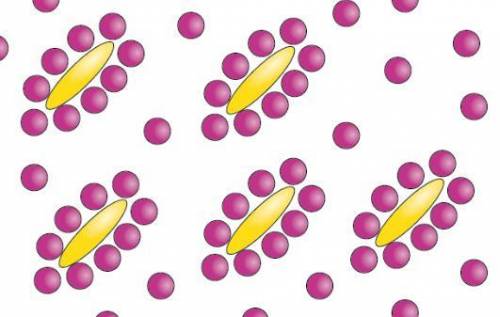
Behold a mixture of oily/hydrophobic (yellow) and water (purplish) molecules. recall that hydrophobic regions are ones with few or no partial charges--so there's nothing for water's hydrogen interaction donors or acceptors to 'play with'. so! how many of the water molecules are 'stuck' contacting at least part of a hydrophobic molecule in this arrangement?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

You know the right answer?
Behold a mixture of oily/hydrophobic (yellow) and water (purplish) molecules. recall that hydrophobi...
Questions












History, 06.03.2020 02:10


Mathematics, 06.03.2020 02:10





Mathematics, 06.03.2020 02:11