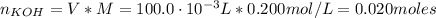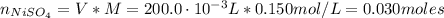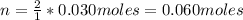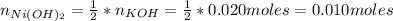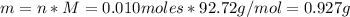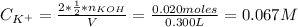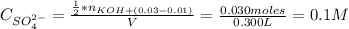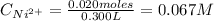A solution of 100.0 mL of 0.200 M KOH is mixed with a solution of 200.0 mL of 0.150 M NiSO4. (a) Write the balanced chemical equation for the reaction that occurs. (b) What precipitate forms? (c) What is the limiting reactant? (d) How many grams of this precipitate form? (e) What is the concentration of each ion that remains in solution?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 23.06.2019 10:00
Why sncl2 is solid while sncl4 is liquid at room temprature explain it in easy way
Answers: 1

Chemistry, 23.06.2019 11:00
Suppose you increase your walking speed from 7 m/s to 15 m/s in a period of 1 s. what is your acceleration?
Answers: 1
You know the right answer?
A solution of 100.0 mL of 0.200 M KOH is mixed with a solution of 200.0 mL of 0.150 M NiSO4. (a) Wri...
Questions


Mathematics, 31.08.2019 04:30



Mathematics, 31.08.2019 04:30


Mathematics, 31.08.2019 04:30


Mathematics, 31.08.2019 04:30

Social Studies, 31.08.2019 04:30

Computers and Technology, 31.08.2019 04:30





Mathematics, 31.08.2019 04:30

Mathematics, 31.08.2019 04:30

History, 31.08.2019 04:30


Mathematics, 31.08.2019 04:30