Chemistry, 02.09.2020 23:01 miguelelmenor910
A protein has an isoelectric point (pI) of 9. Which of the following statements best describes the state of this protein in aqueous solution at pH 7?
A. The side chains of the protein's basic residues will be mostly uncharged
B. The side chains of the protein's acidic residues will be mostly protonated
C. The protein will have no net charge
D. The protein will have a net positive charge
E. The protein will have a net negative charge

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3

Chemistry, 23.06.2019 01:30
Polar bears give birth and hunt on sea ice. which of the following would polar bears survive during the melting of arctic ice? growing another layer of fur during summer migrate inland to search for different food sources staying put until the ice refreezes sticking to the usual diet of seals
Answers: 1

Chemistry, 23.06.2019 06:30
What is the chemical formula for a compound between li and br? libr li2br libr2 libr3
Answers: 1
You know the right answer?
A protein has an isoelectric point (pI) of 9. Which of the following statements best describes the s...
Questions

Computers and Technology, 29.01.2021 01:00




Mathematics, 29.01.2021 01:00

Mathematics, 29.01.2021 01:00


Mathematics, 29.01.2021 01:00


History, 29.01.2021 01:00



Mathematics, 29.01.2021 01:00

English, 29.01.2021 01:00

Biology, 29.01.2021 01:00


Chemistry, 29.01.2021 01:00

Mathematics, 29.01.2021 01:00

Mathematics, 29.01.2021 01:00

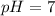
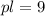
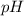 is below
is below