A solution is made by dissolving
37.5 g of sodium sulfide (Na2S) in
217 g of water.
Wha...

Chemistry, 03.09.2020 21:01 ilovecatsomuchlolol
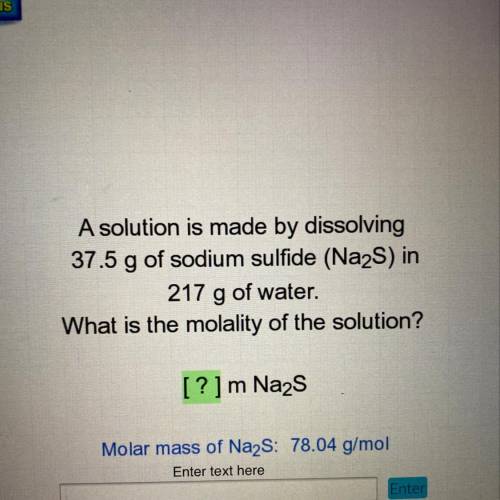
A solution is made by dissolving
37.5 g of sodium sulfide (Na2S) in
217 g of water.
What is the molality of the solution?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
Questions


English, 04.10.2019 20:50




English, 04.10.2019 20:50



English, 04.10.2019 20:50


Biology, 04.10.2019 20:50

History, 04.10.2019 20:50


Mathematics, 04.10.2019 20:50

Physics, 04.10.2019 20:50

Biology, 04.10.2019 20:50






