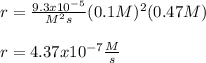The reaction 2NO(g) + O2(g) 2NO2(g) is second order in NO and first order in O2. When [NO] = 0.8 M and [O2] = 3.7 M, the observed rate of the reaction is 0.00022022 M/s. (a) What is the value of the rate constant? (d) What is the rate of reaction when [NO] = 0.1 M and [O2] = 0.47 M?

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1

Chemistry, 23.06.2019 03:20
High-pressure liquid chromatography (hplc) is a method used in chemistry and biochemistry to purify chemical substances. the pressures used in this procedure range from around 500 kilopascals (500,000 pa) to about 60,000 kpa (60,000,000 pa). it is often convenient to know the pressure in torr. if an hplc procedure is running at a pressure of 1.03×108 pa , what is its running pressure in torr?
Answers: 3

Chemistry, 23.06.2019 04:20
The lewis diagrams for magnesium and fluorine are shown below. what is the correct chemical formula for magnesium fluoride? a. mgf b. mg2f c. mgf2 d. mg2f3
Answers: 1
You know the right answer?
The reaction 2NO(g) + O2(g) 2NO2(g) is second order in NO and first order in O2. When [NO] = 0.8 M a...
Questions

Social Studies, 28.01.2020 14:53

Mathematics, 28.01.2020 14:53

Computers and Technology, 28.01.2020 14:53



History, 28.01.2020 14:53

History, 28.01.2020 14:53

History, 28.01.2020 14:53


Biology, 28.01.2020 14:53

Mathematics, 28.01.2020 14:53


History, 28.01.2020 14:53

Mathematics, 28.01.2020 14:53

Mathematics, 28.01.2020 14:53





World Languages, 28.01.2020 14:53

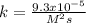
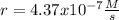
![r=k[NO]^2[O_2]](/tpl/images/0747/3983/b9fab.png)
![k=\frac{r}{[NO]^2[O_2]}\\\\k=\frac{0.00022022M/s}{(0.8M)^2(3.7M)} \\\\k=\frac{9.3x10^{-5}}{M^2s}](/tpl/images/0747/3983/c4bb6.png)