
Chemistry, 10.09.2020 03:01 CoreyHammond1517
When 229.0 J of energy is supplied as heat to 3.00 mol of Ar(g) at constant pressure the temperature of the sample increases by 2.55 K. Assuming that in the experiment the gas behaves as an ideal gas, calculate the molar heat capacities at constant volume and at constant pressure of Ar(g).

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 04:30
This question is about electrolysis. metal spoons can be coated with silver. this is called electroplating. suggest one reason why spoons are electroplated?
Answers: 1

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

You know the right answer?
When 229.0 J of energy is supplied as heat to 3.00 mol of Ar(g) at constant pressure the temperature...
Questions


Mathematics, 03.02.2021 08:10


English, 03.02.2021 08:10




Mathematics, 03.02.2021 08:10

Mathematics, 03.02.2021 08:10


Mathematics, 03.02.2021 08:10






Business, 03.02.2021 08:10

Biology, 03.02.2021 08:10

Physics, 03.02.2021 08:10

Mathematics, 03.02.2021 08:10

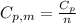
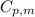 is the molar heat capacity at constant pressure
is the molar heat capacity at constant pressure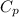 is the heat capacity at constant pressure
is the heat capacity at constant pressure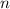 is the number of moles
is the number of moles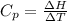
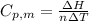
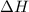 = 229.0 J
= 229.0 J = 2.55 K
= 2.55 K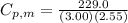
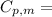 29.93 JK⁻¹mol⁻¹
29.93 JK⁻¹mol⁻¹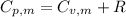
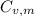 is the molar heat capacity at constant volume
is the molar heat capacity at constant volume 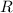 is the gas constant (
is the gas constant (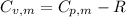
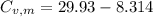
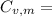 21.62 JK⁻¹mol⁻¹
21.62 JK⁻¹mol⁻¹

