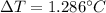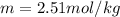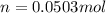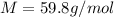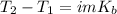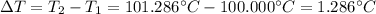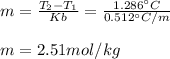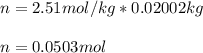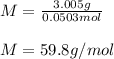Chemistry, 20.09.2020 15:01 bradleydb222
Suppose 3.005 g of a nonvolatile solute is added to 20.02 g of water (the solvent), and the boiling point increases from 100.000 OC to 101.286 OC. Determine the TB, molality, moles, and molecular weight for the solute if kb for water is 0.512 OC/m. Report each value using the correct number of significant digits. Refer to Example 1.2 and pages 3-4 in the chapter 1 notes for general chemistry 1 to understand significant figures. Also, include all applicable units and conversion factors.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 18:00
Alidded glass container is filled with a colored gas. after a period of time, it is observed that the gas is uniformly spread throughout the box and that the movement has slowed considerably. next, a warm iron plate is carefully placed under the box. why is there resumed movement of the gas in the container?
Answers: 2

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3
You know the right answer?
Suppose 3.005 g of a nonvolatile solute is added to 20.02 g of water (the solvent), and the boiling...
Questions



Mathematics, 30.10.2019 10:31



Spanish, 30.10.2019 10:31

History, 30.10.2019 10:31




Mathematics, 30.10.2019 10:31

Physics, 30.10.2019 10:31


History, 30.10.2019 10:31

English, 30.10.2019 10:31



Chemistry, 30.10.2019 10:31

Mathematics, 30.10.2019 10:31

Chemistry, 30.10.2019 10:31