
Chemistry, 22.09.2020 05:01 haleylayne74
A nearly flat bicycle tire becomes noticeably warmer after it has been pumped up. Approximate this process as a reversible adiabatic compression. Take the initial pressure and temperature of the air before it is put in the tire to be Pi = 1.00 bar and Ti = 298 K. The final volume of the air in the tire is Vf= 1.50 L and the final pressure is Pf = 5.00 bar. Calculate the final temperature of the air in the tire.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 12:30
4nh3+5o2--> 4no+6h20what is the total number of moles of h2o produced when 12 mole of nh3 is completely consumed?
Answers: 3

Chemistry, 22.06.2019 01:30
There are main groups in the modern periodic table of elements
Answers: 1

Chemistry, 22.06.2019 06:00
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

You know the right answer?
A nearly flat bicycle tire becomes noticeably warmer after it has been pumped up. Approximate this p...
Questions

Biology, 22.09.2019 21:00

Mathematics, 22.09.2019 21:00


English, 22.09.2019 21:00

Business, 22.09.2019 21:00


Mathematics, 22.09.2019 21:00

History, 22.09.2019 21:00

Mathematics, 22.09.2019 21:00

English, 22.09.2019 21:00

History, 22.09.2019 21:00

Mathematics, 22.09.2019 21:00


Chemistry, 22.09.2019 21:00

Mathematics, 22.09.2019 21:00



Mathematics, 22.09.2019 21:00

Mathematics, 22.09.2019 21:00

History, 22.09.2019 21:00

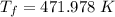
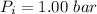
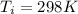
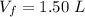
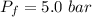
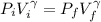
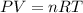
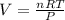
![P_i *[ \frac{n RT_i}{P_i}]^{\gamma} = P_f [ \frac{n RT_f}{P_f}]^{\gamma}](/tpl/images/0775/6605/1dfe6.png)
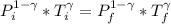
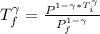
![T_f =[ \frac{ P_i ^{1 - \gamma} * T_i^{\gamma}}{P_f^{1 - \gamma}}]^{\frac{1}{\gamma} }](/tpl/images/0775/6605/333d0.png)
![T_f = T_i * [\frac{P_i}{P_f} ]^{\frac{1- \gamma}{\gamma}](/tpl/images/0775/6605/2faae.png)
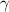 is a constant mathematically represented as
is a constant mathematically represented as 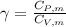
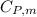 is the molar heat capacity at constant pressure
is the molar heat capacity at constant pressure 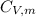 is the molar heat capacity at constant volume given as
is the molar heat capacity at constant volume given as 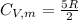
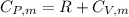
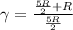
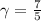
![T_f = T_i * [\frac{P_i}{P_f} ]^{\frac{1- \frac{7}{5}}{ \frac{7}{5}}](/tpl/images/0775/6605/9c28a.png)
![T_f = 298 * [\frac{1}{5} ]^{\frac{1- \frac{7}{5}}{ \frac{7}{5}}](/tpl/images/0775/6605/9e54c.png)


