Chemistry, 22.09.2020 21:01 briannasineiro7937
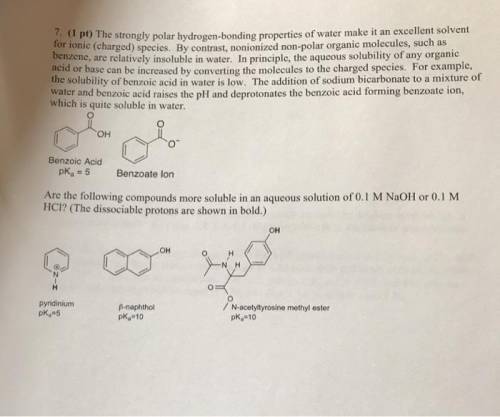
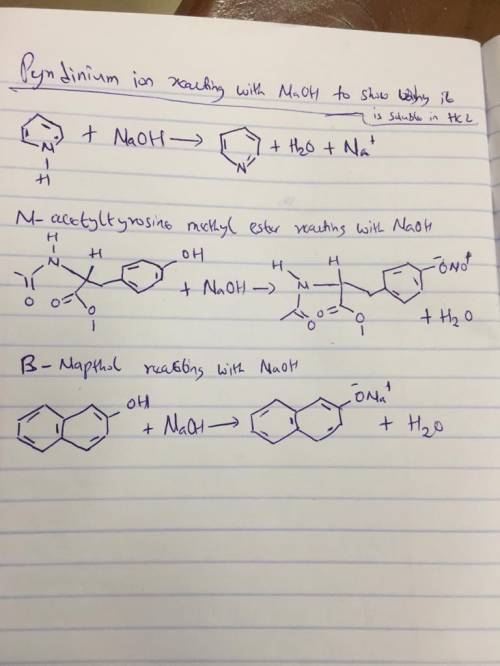
The strongly polar, hydrogen-bonding properties of water make it an excellent solvent for ionic (charged) species. By contrast, nonionized, nonpolar organic molecules, such as benzene, are relatively insoluble in water. In principle, the aqueous solubility of any organic acid or base can be increased by converting the molecules to charged species. For example, the solubility of benzoic acid in water is low. The addition of sodium bicarbonate to a mixture of water and benzoic acid raises the pH and deprotonates the benzoic acid to form benzoate ion, which is quite soluble in water?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1
You know the right answer?
The strongly polar, hydrogen-bonding properties of water make it an excellent solvent for ionic (cha...
Questions


Mathematics, 28.01.2021 03:40

Mathematics, 28.01.2021 03:40

Mathematics, 28.01.2021 03:40

Mathematics, 28.01.2021 03:40




English, 28.01.2021 03:40


History, 28.01.2021 03:40

History, 28.01.2021 03:40


Mathematics, 28.01.2021 03:40

Chemistry, 28.01.2021 03:40


Chemistry, 28.01.2021 03:40

English, 28.01.2021 03:40


Social Studies, 28.01.2021 03:40