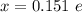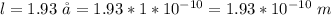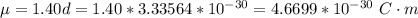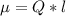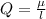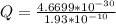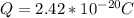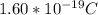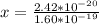Chemistry, 23.09.2020 14:01 solikhalifeoy3j1r
Determine the partial negative charge on the bromine atom in a c−br bond. the bond length is 1.93 å and the bond dipole moment is 1.40 d . express your answer using 3 significant figures. the partial negative charge on the bromine atom = previous answersrequest answer incorrect; try again; 4 attempts remaining provide feedback.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1

Chemistry, 22.06.2019 16:00
Uranium can supply energy for the worlds electricity without admitting harmful greenhouse gases which of these statements best describes an outcome of uranium mining
Answers: 1

Chemistry, 22.06.2019 17:20
Pegmatites are igneous rocks in which the individual minerals are very large. typically, the minerals are all light-colored quartz, feldspar and muscovite. if you were given a black and white photograph of a pegmatite in a quarry (where the rock has been blasted and broken), what physical properties could you use to identify those three minerals in this hypothetical photo? describe each mineral and the specific diagnostic properties. be specific.
Answers: 2

Chemistry, 22.06.2019 20:00
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3
You know the right answer?
Determine the partial negative charge on the bromine atom in a c−br bond. the bond length is 1.93 å...
Questions

English, 02.02.2021 05:30




Mathematics, 02.02.2021 05:30

English, 02.02.2021 05:30




Mathematics, 02.02.2021 05:30


Mathematics, 02.02.2021 05:30





Social Studies, 02.02.2021 05:30

Mathematics, 02.02.2021 05:30