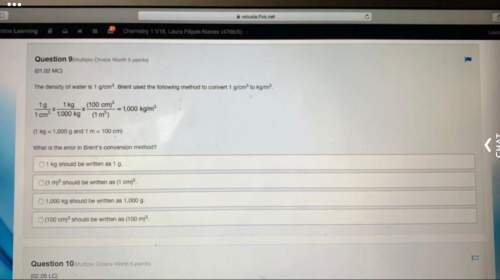
The characteristics of two unknown intermolecular forces, C and D, are compared in the table.
Intermolecular Forces
CD
Occurs between polar asymmetrical moleculesOccurs between non-polar or polar symmetric molecules
Which of the following conclusions about the two intermolecular forces is correct? (1 point)
a
C represents ion-dipole forces, and D represents dipole-dipole forces
b
C represents dipole-dipole forces, and D represents London dispersion forces
c
C represents London dispersion forces, and D represents ion-dipole forces
d
C represents hydrogen bonding, and D represents London dispersion forces

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1


Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

You know the right answer?
The characteristics of two unknown intermolecular forces, C and D, are compared in the table.
Inter...
Questions

History, 29.05.2020 21:59


English, 29.05.2020 21:59


History, 29.05.2020 21:59

Health, 29.05.2020 21:59

Mathematics, 29.05.2020 21:59

Physics, 29.05.2020 21:59

Mathematics, 29.05.2020 21:59


Mathematics, 29.05.2020 21:59

Mathematics, 29.05.2020 21:59

English, 29.05.2020 21:59


Mathematics, 29.05.2020 21:59