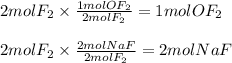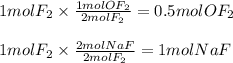A 2 mole sample of F2(g) reacts with excess NaOH(aq) according to the equation above. If the reaction is repeated with excess NaOH(aq) but with 1 mole of F2(g), which of the following is correct?
Group of answer choices
The amount of OF2(g) produced is doubled.
The amount of OF2(g) produced is halved.
The amount of NaF(aq) produced remains the same.
The amount of NaF(aq) produced is doubled.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1


You know the right answer?
A 2 mole sample of F2(g) reacts with excess NaOH(aq) according to the equation above. If the reactio...
Questions


Biology, 23.04.2020 21:22

Geography, 23.04.2020 21:22


French, 23.04.2020 21:22



Chemistry, 23.04.2020 21:22

Mathematics, 23.04.2020 21:22

Computers and Technology, 23.04.2020 21:22

History, 23.04.2020 21:22



Mathematics, 23.04.2020 21:22

Spanish, 23.04.2020 21:22

English, 23.04.2020 21:22

Mathematics, 23.04.2020 21:23


Biology, 23.04.2020 21:23