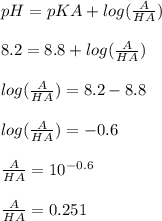Chemistry, 24.09.2020 14:01 because061907
A compound is known to have a free amino group with a pKa of 8.8, and one other ionizable group with a pKa between 5 and 7. To 100 mL of a 0.2 M solution of this compound at pH 8.2 was added 40 mL of a solution of 0.2 M hydrochloric acid. The pH changed to 6.2. The pKa of the second ionizable group is: A) The pH cannot be determined from this information. B) 5.4. C) 5.6. D) 6.0. E) 6.2.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 00:30
Maya wrote if you step to describe how carbon circulates between the atmosphere and living organisms
Answers: 1

You know the right answer?
A compound is known to have a free amino group with a pKa of 8.8, and one other ionizable group with...
Questions







Mathematics, 06.05.2020 03:09






Mathematics, 06.05.2020 03:09






Mathematics, 06.05.2020 03:09