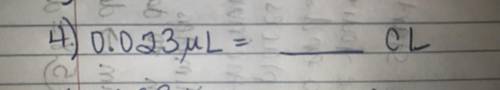HELP I WILL MARK YOU BRAINLIEST
0.023 ml= blank cl
...

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
Questions

History, 21.01.2021 23:30



Mathematics, 21.01.2021 23:30



Mathematics, 21.01.2021 23:30


Mathematics, 21.01.2021 23:30



English, 21.01.2021 23:30


Computers and Technology, 21.01.2021 23:30

History, 21.01.2021 23:30

Chemistry, 21.01.2021 23:30


Mathematics, 21.01.2021 23:30

History, 21.01.2021 23:30

Geography, 21.01.2021 23:30