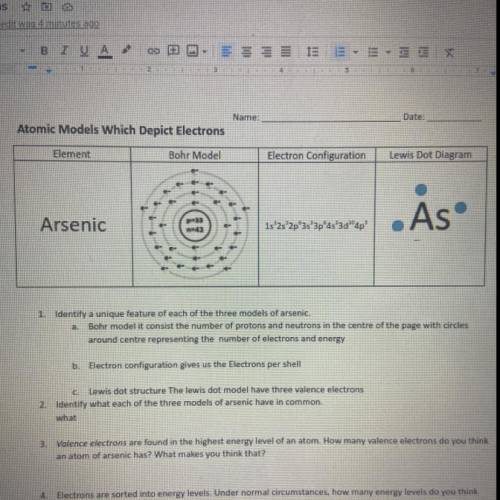
Atomic Models Which Depict Electrons
Element
Bohr Model
Electron Configuration
Le...

Chemistry, 29.09.2020 20:01 queenkendra16
Atomic Models Which Depict Electrons
Element
Bohr Model
Electron Configuration
Lewis Dot Diagram
. As
'
Arsenic
1s 2s 2p 3s 3pas'3d"
ap'
1. Identify a unique feature of each of the three models of arsenie.
Bohr modelit consist the number of protons and neutrons in the centre of the page with circles
around contre representing number of electrons and energy
a
D: Electron configuration gives us the Electrons per shell
Lewis dot structure The lewis dot model have three valence electrons
2. Identify what each of the three models of arsenic have in common
what
Valence electrons are found in the highest energy level of an atom. How many valence electrons do you think
an atom of arsenic has? What makes you think that?
4. Electrons are sorted into energy levels. Under normal circumstances, how many energy levels do you think
contain electrons in an atom of arsenic? What makes you think that?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1


Chemistry, 23.06.2019 08:40
Calculate the number of grams of sodium in 3.00 g of each sodium-containing food additive.
Answers: 3
You know the right answer?
Questions


English, 10.03.2021 01:50


Mathematics, 10.03.2021 01:50




Mathematics, 10.03.2021 01:50


Health, 10.03.2021 01:50


Mathematics, 10.03.2021 01:50


Mathematics, 10.03.2021 01:50

Mathematics, 10.03.2021 01:50


English, 10.03.2021 01:50

Physics, 10.03.2021 01:50

History, 10.03.2021 01:50




