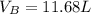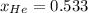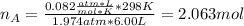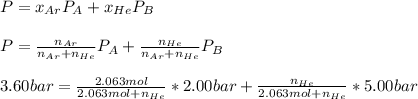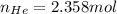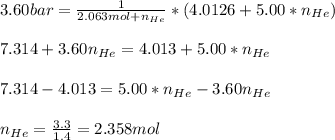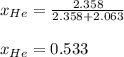Chemistry, 04.10.2020 17:01 devenybates
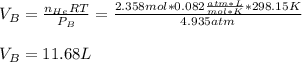
Argon (Ar) and helium (He) are initially in separate compartments of a container at 25°C. The
Ar in compartment A, which has a volume VA of 6.00 L, has a pressure of 2.00 bar. The He in
compartment B of unknown volume V3 has a pressure of 5.00 bar. When the two compartments
are connected and the gases allowed to mix, the total pressure of gas is 3.60 bar. Assume both
gases behave ideally
(a) [4 marks) Determine the volume of compartment B.
(b) [2 marks] Determine the mole fraction of He in the mixture of gases.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

You know the right answer?
Argon (Ar) and helium (He) are initially in separate compartments of a container at 25°C. The
Ar in...
Questions

Health, 21.09.2019 20:30


English, 21.09.2019 20:30

Biology, 21.09.2019 20:30

Mathematics, 21.09.2019 20:30

History, 21.09.2019 20:30

Mathematics, 21.09.2019 20:30

History, 21.09.2019 20:30


Physics, 21.09.2019 20:30



Computers and Technology, 21.09.2019 20:30

History, 21.09.2019 20:30

Mathematics, 21.09.2019 20:30


Geography, 21.09.2019 20:30