
Terry bought a gold necklace at a pawnshop and was wondering if it was “real” gold. She decided to perform a density test. She placed the necklace in a graduated cylinder of water that had 47.00 mL of water and the water rose to 48.35 mL. She then weighed the necklace and found it to have a mass of 22.89 g. What is the density of Terry’s necklace?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1

Chemistry, 23.06.2019 01:30
What is produced from neutralization of an acid and a base? a. hydronium ions b. citric acid c. salt and water
Answers: 1
You know the right answer?
Terry bought a gold necklace at a pawnshop and was wondering if it was “real” gold. She decided to p...
Questions

Mathematics, 18.09.2019 01:00


Mathematics, 18.09.2019 01:00

History, 18.09.2019 01:00


Mathematics, 18.09.2019 01:00

History, 18.09.2019 01:00

English, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00

Biology, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00


Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00


Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00

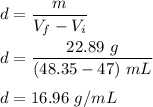 '
'

