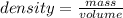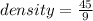Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 11:00
Suppose you increase your walking speed from 7 m/s to 15 m/s in a period of 1 s. what is your acceleration?
Answers: 1
You know the right answer?
If an object has a mass of 45g and a volume of 9 cubic centimeters, what is its density? been wrokin...
Questions

Physics, 07.12.2021 01:00

History, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00





Health, 07.12.2021 01:00

History, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00


History, 07.12.2021 01:00

English, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00



Chemistry, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00

Mathematics, 07.12.2021 01:00