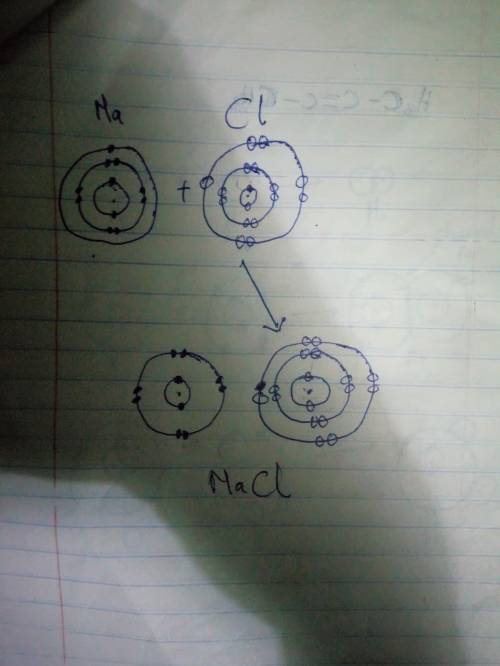
Chemistry, 07.10.2020 08:01 avadestinyy
The following figure represents the formation of an ionic compound. Substances A and B are initially uncharged, but when mixed electrons are transferred. Using the figure, identify which substance will form the cation and which will form the anion. Provide a brief (one or two sentences) explanation for your response. (Hint: How does losing electrons affect atomic radii?)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Describe the chemical reaction based on the chemical equation below. also, explain whether the equation is balanced.
Answers: 1

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1

Chemistry, 23.06.2019 08:10
An experiment is conducted to see if cats preferred skim milk or 2% milk. a cup of skim milkwas put out for 5 kittens and then measured how much the kittens drank over the course of aday. following a cup of 2% milk was purout for the skittens and then masured how much thekittens drank over the course of a day. the same kittens were used and the milk was served atthe same temperature. it was discovered that the cats liked the 2% milk more than the skimmilk. what is the dependent variable in this experiment?
Answers: 1
You know the right answer?
The following figure represents the formation of an ionic compound. Substances A and B are initially...
Questions



Mathematics, 18.10.2021 17:50



Social Studies, 18.10.2021 17:50


History, 18.10.2021 17:50


Physics, 18.10.2021 17:50

Mathematics, 18.10.2021 17:50

Mathematics, 18.10.2021 17:50


Mathematics, 18.10.2021 17:50

Mathematics, 18.10.2021 17:50

Mathematics, 18.10.2021 17:50

Mathematics, 18.10.2021 17:50