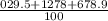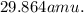Chemistry, 08.10.2020 08:01 snowprincess99447
3. What is the atomic mass of phosphorous if phosphorous-29 has a percent abundance of 35.5%, phosphorous-30 has a percent abundance of 42.6%, and phosphorous-31 has a percent abundance of 21.9%?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 23:00
What is the mass of naoh that would have to be added to 500 ml of a solution of 0.20 m acetic acid in order to achieve a ph of 5.0?
Answers: 1

Chemistry, 23.06.2019 01:30
List and describe the neurological effects of the vocs and other air pollutants,as described by dr.theo colborn
Answers: 2

Chemistry, 23.06.2019 02:30
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1

Chemistry, 23.06.2019 09:00
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate according to the following equation:
Answers: 2
You know the right answer?
3. What is the atomic mass of phosphorous if phosphorous-29 has a percent abundance of 35.5%, phosph...
Questions

Mathematics, 01.12.2021 05:10


Mathematics, 01.12.2021 05:10

Chemistry, 01.12.2021 05:10

Mathematics, 01.12.2021 05:10


English, 01.12.2021 05:10


Mathematics, 01.12.2021 05:10

Mathematics, 01.12.2021 05:10

Biology, 01.12.2021 05:10

Geography, 01.12.2021 05:10

Mathematics, 01.12.2021 05:10

Law, 01.12.2021 05:10

Mathematics, 01.12.2021 05:10

History, 01.12.2021 05:10

Mathematics, 01.12.2021 05:10


Social Studies, 01.12.2021 05:10