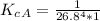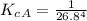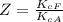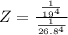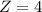Chemistry, 09.10.2020 23:01 andybiersack154
The protein hemoglobin (Hb) transports O2 in mammalian blood. Each Hb can bind 4O2 molecules. The equilibrium constant for the O2-binding reaction is higher in fetal hemoglobin than in adult hemoglobin. In discussing protein oxygen-binding capacity, biochemists use a measure called the P50 value, defined as the partial pressure of oxygen at which 50% of the protein is saturated. Fetal hemoglobin has a P50 value of 19 torr, and adult hemoglobin has a P50 value of 26.8 torr. Use these data to estimate how much larger Kc is for the aqueous reaction 4O2(g)+Hb(aq)→[Hb(O2)4(aq)].

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1

Chemistry, 22.06.2019 10:30
Earth's axis of rotation is tilted at an angle of 23.5 degrees. what is one change you would see on earth if its axis was not tilted?
Answers: 3

Chemistry, 22.06.2019 12:00
the mississippians were considered to be horticulturalists, which means they were
Answers: 1
You know the right answer?
The protein hemoglobin (Hb) transports O2 in mammalian blood. Each Hb can bind 4O2 molecules. The eq...
Questions

Mathematics, 13.01.2020 10:31




Biology, 13.01.2020 10:31

History, 13.01.2020 10:31


Social Studies, 13.01.2020 10:31


Mathematics, 13.01.2020 10:31


Mathematics, 13.01.2020 10:31

Chemistry, 13.01.2020 10:31




Mathematics, 13.01.2020 10:31

Mathematics, 13.01.2020 10:31

Mathematics, 13.01.2020 10:31

English, 13.01.2020 10:31

![[P_{O_2}]_F = 19 torr](/tpl/images/0795/3120/d5cf5.png)
![[P_{O_2}]_A = 26.8 torr](/tpl/images/0795/3120/e335c.png)
![4O_2_{(g)}+Hb_{(aq)}\to [Hb(O_2)_4_{{(aq)}}]](/tpl/images/0795/3120/45f03.png)
![K_c_F = \frac{[P_{[Hb(O_2)_4}]}{ [P_{O_2}]_F^4 * [P_{Hb}]}](/tpl/images/0795/3120/f4794.png)
![[P_{[Hb(O_2)_4}] [\tex] and [P_{Hb}] will be 1 because both substances are aqueousSo [tex]K_c_F = \frac{1}{ 19^4 *1 }](/tpl/images/0795/3120/59557.png)
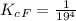
![K_c_A = \frac{[P_{[Hb(O_2)_4}]}{ [P_{O_2}]_A^4 * [P_{Hb}]}](/tpl/images/0795/3120/10577.png)