Chemistry, 11.10.2020 23:01 hhomeschool24
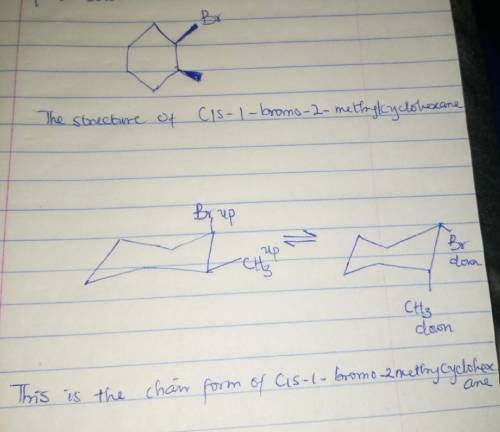
The two alternative chair conformations of cis-1-bromo-2-methylcyclohexane differ in their Gibbs free energy. Using the data for ΔG (Axial-Equatorial) for monosubstituted cyclohexanes at room temperature (25ºC): Axial → Equatorial Group ΔG° (kJ/mol) Group ΔG° (kJ/mol) -0.8 -5.9 -2.4 -7.3 -3.9 1,2-gauche 3.8 Calculate the absolute value of the difference in the Gibbs free energy between the alternative chair conformations. kJ/mol Which group in this compound is in axial position in the energetically preferred chair conformation?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

You know the right answer?
The two alternative chair conformations of cis-1-bromo-2-methylcyclohexane differ in their Gibbs fre...
Questions

Biology, 16.07.2019 06:30


Biology, 16.07.2019 06:30



History, 16.07.2019 06:30

History, 16.07.2019 06:30




Mathematics, 16.07.2019 06:30


Health, 16.07.2019 06:30

History, 16.07.2019 06:30

Physics, 16.07.2019 06:30

Social Studies, 16.07.2019 06:30

History, 16.07.2019 06:30

Social Studies, 16.07.2019 06:30