
Chemistry, 12.10.2020 22:01 noahdeno200010125
How do I do this question?The aluminum cup inside your calorimeter weighs 39.78 g. You add 50.01 g of ice cold water to the calorimeter. You measure the temperature of the calorimeter to be 0.5oC just before your next addition. You then add 50.72 g of hot water and a 49.98 g metal object, all having an initial temperature of 69.5oC. After the calorimeter reaches thermal equilibrium, the final temperature is measured to be 35.9oC.
What is the specific heat of the metal object, in units of J g-1 oC-1.
Assume that:
the calorimeter is completely insulated
the heat capacity of the empty calorimeter is the heat capacity of the aluminum cup: 0.903 J g-1 oC-1.
the density of water is: 1.00 g/mL.
the heat capacity of water is: 4.184 J g-1 oC-1.
Perform all calculations without rounding, but then provide your answer to the correct number of significant figures. Units can be entered as J/(gK)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Consider three unlabeled bottles, each contain small pieces of one of the following metals. - magnesium - sodium - silver the following reagents are used for identifying the metals. - pure water - a solution of 1.0 molar hcl - a solution of concentrated hno3 (a) which metal can be easily identified because it is much softer than the other two? describe a chemical test that distinguishes this metal from the other two, using only one of the reagents above. write a balanced chemical equation for the reaction that occurs. (b) one of the other two metals reacts readily with the hcl solution. identify the metal and write the balanced chemical equation for the reaction that occurs when this metal is added to the hcl solution. use the table of standard reduction potentials (attached) to account for the fact that this metal reacts with hcl while the other does not. (c) the one remaining metal reacts with the concentrated hno3 solution. write a balanced chemical equation for the reaction that occurs. (d) the solution obtained in (c) is diluted and a few drops of 1 m hcl is added. describe what would be observed. write a balanced chemical equation for the reaction that occurs.
Answers: 2

Chemistry, 22.06.2019 21:00
Two nails have identical sizes and shapes. in one nail, 20 percent of the domains are lined up. in the other nail, 80 percent of the domains are lined up. which has stronger magnetic force? first answer gets brainliest!
Answers: 1

Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1

Chemistry, 23.06.2019 07:00
What are the trends and exceptions to the trends in electron affinity?
Answers: 1
You know the right answer?
How do I do this question?The aluminum cup inside your calorimeter weighs 39.78 g. You add 50.01 g o...
Questions



Chemistry, 20.05.2021 16:30

Mathematics, 20.05.2021 16:30





Mathematics, 20.05.2021 16:30

Computers and Technology, 20.05.2021 16:30



English, 20.05.2021 16:30

Mathematics, 20.05.2021 16:30

Mathematics, 20.05.2021 16:30

Mathematics, 20.05.2021 16:30

Mathematics, 20.05.2021 16:30



History, 20.05.2021 16:30

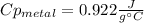
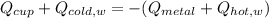
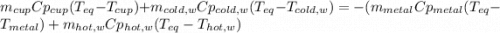
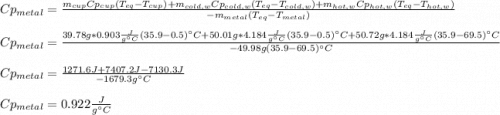 Best regards.
Best regards.

