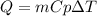Chemistry, 13.10.2020 03:01 igivehead666
You found that the unknown metal has a specific heat capacity close to, for example, that of silver [c=234J/(kg⋅K)]. Let's compare silver to other known materials and see whether your results make sense. Imagine you have 1 kg of each of the substances listed below. If the same amount of heat Q is added to each, which substance will undergo the largest change in temperature ΔT? Assume that no substance experiences a phase change. Rank the samples from largest to smallest change in temperature.
a. c= silver/ 234J(kg/K)
b. c= water/ 4190 J(kg/K)
c. c= glass/ 754J/(kg. K)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3
You know the right answer?
You found that the unknown metal has a specific heat capacity close to, for example, that of silver...
Questions



Mathematics, 27.05.2020 21:00


Mathematics, 27.05.2020 21:00



Mathematics, 27.05.2020 21:00




Mathematics, 27.05.2020 21:00

Mathematics, 27.05.2020 21:01


History, 27.05.2020 21:01



History, 27.05.2020 21:01