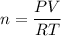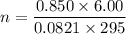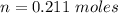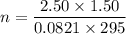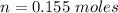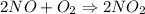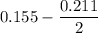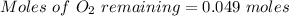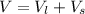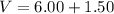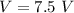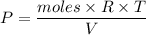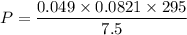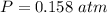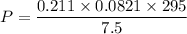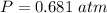Two bulbs are connected by a stopcock. The large bulb, with a volume of 6.00 L, contains nitric oxide at a pressure of 0.850 atm, and the small bulb, with a volume of 1.50 L, contains oxygen at a pressure of 2.50 atm. The temperature at the beginning and the end of the experiment is 22∘C .
After the stopcock is opened, the gases mix and react.
2NO(g)+O2(g)→2NO2(g)
1. Which gases are present at the end of the experiment?
2. What are the partial pressures of the gases? If the gas was consumed completely, put 0 for the answer.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2
You know the right answer?
Two bulbs are connected by a stopcock. The large bulb, with a volume of 6.00 L, contains nitric oxid...
Questions



Computers and Technology, 21.09.2020 08:01

History, 21.09.2020 08:01

Mathematics, 21.09.2020 08:01






Mathematics, 21.09.2020 08:01

Mathematics, 21.09.2020 08:01


Mathematics, 21.09.2020 08:01

Mathematics, 21.09.2020 08:01

Arts, 21.09.2020 08:01




Mathematics, 21.09.2020 08:01