
Chemistry, 16.10.2020 18:01 samarth4315
What mass of the calcium carbonate, to the nearest hundredth of a gram, is decomposed in this reaction.
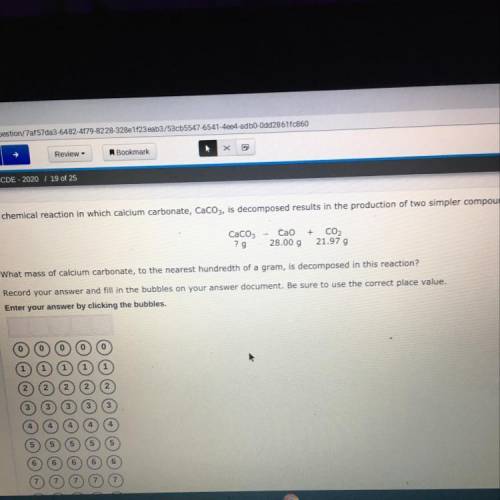

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 23.06.2019 08:30
Benzonitrile (c6h5cn) is reduced to two different products depending on the reducing agent used. treatment with lithium aluminum hydride followed by water forms k, which has a molecular ion in its mass spectrum at 107 and the following ir absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. treatment with a milder reducing agent forms l, which has a molecular ion in its mass spectrum at 106 and the following ir absorptions: 3086, 2850, 2820, 2736, 1703, and 1600 cm-1. l shows fragments in its mass spectrum at m/z = 105 and 77. propose structures for k and l and choose an explanation for how this could be concluded.
Answers: 3

Chemistry, 23.06.2019 15:00
This is a portion of the earths surface that may be far from tectonic plates boundaries yet experiences volcanism due to a rising mantle plume or some other cause
Answers: 3

Chemistry, 23.06.2019 16:50
An element x has two isotopes. x–15 has 7 protons and 8 neutrons. determine the atomic number and mass number of the second isotope, which has 10 neutrons in its nucleus.
Answers: 1
You know the right answer?
What mass of the calcium carbonate, to the nearest hundredth of a gram, is decomposed in this reacti...
Questions


Mathematics, 14.10.2019 09:30

Mathematics, 14.10.2019 09:30

Mathematics, 14.10.2019 09:30


Mathematics, 14.10.2019 09:30



Mathematics, 14.10.2019 09:30


History, 14.10.2019 09:30

Mathematics, 14.10.2019 09:30



History, 14.10.2019 09:30


Social Studies, 14.10.2019 09:30


English, 14.10.2019 09:30



