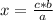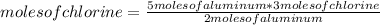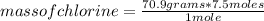Chemistry, 17.10.2020 06:01 GreenHerbz206
Aluminum metal reacts with chlorine gas. Determine the mass of chlorine to react completely with 5.0 moles of aluminum.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
The overall chemical reaction for photosynthesis isshown below: 6co2 + 6h20 → c6h12o6 + 602what mass of glucose (c6h1206) can form from71.89 g co2? (molar mass of c6h1206 = 180.18g/mol; molar mass of co2 = 44.01 g/mol)71.89 g co2=g c6h1206
Answers: 1

Chemistry, 21.06.2019 20:30
If 10.g of agno3 is available, what volume of 0.25 m agno3 can be prepared
Answers: 1

Chemistry, 22.06.2019 04:20
Neils bohr believed that electrons orbited the nucleus in different energy levels, based on strong support from
Answers: 1

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
You know the right answer?
Aluminum metal reacts with chlorine gas. Determine the mass of chlorine to react completely with 5.0...
Questions


History, 16.01.2020 02:31


Mathematics, 16.01.2020 02:31

Mathematics, 16.01.2020 02:31

Computers and Technology, 16.01.2020 02:31




Biology, 16.01.2020 02:31



Mathematics, 16.01.2020 02:31







History, 16.01.2020 02:31