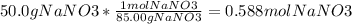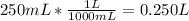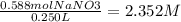Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 23.06.2019 04:10
In an experiment, 45g of silicon tetrachloride are treated with 45ml of water. what is the theoretical yield in grams of hcl
Answers: 3

Chemistry, 23.06.2019 07:40
Which of the following has expanded our knowledge of the universe beyond our solar system the most? a. manned space travel b. the hubble space telescope c. the pioneer and voyager missions d. the international space station
Answers: 3
You know the right answer?
50.0 g of NaNO3 are dissolved into enough water to make 250 mL of solution. What is the molarity of...
Questions

Mathematics, 14.07.2019 08:30

Mathematics, 14.07.2019 08:30

History, 14.07.2019 08:30


Mathematics, 14.07.2019 08:30

Mathematics, 14.07.2019 08:30

Mathematics, 14.07.2019 08:30





History, 14.07.2019 08:30


Mathematics, 14.07.2019 08:30


Mathematics, 14.07.2019 08:30


Mathematics, 14.07.2019 08:30

Mathematics, 14.07.2019 08:30

Mathematics, 14.07.2019 08:30